|
Chondroitin is a
widely used ingredient that occurs naturally in the body of
warm-blooded animals, and forms an important building block of joint
cartilage. Dozens of scientific studies with animals and humans have
clearly demonstrated that dietary supplementation of chondroitin in
combination with Glucosamine, another building block of cartilage,
benefits joints. The best-known clinical study is the 4-year one
known as the Glucosamine/chondroitin Arthritis Intervention Trial
(GAIT), conducted at 16 sites across the United States. This
double-blind, placebo-controlled study was sponsored by the
government funded National Institute of Health (NIH), and was
published in 2005 in the New England Journal of Medicine. This
study, which enrolled nearly 1,600 patients, showed that when
Glucosamine and Chondroitin were used in combination, they resulted
in relief of knee pain in nearly 79% of patients with moderate to
severe osteoarthritis; significantly more than either the placebo
control group, or patients that were treated with the common NSAID
drug Celecoxib (Celebrex).
The chondroitin
sulfate offered to manufacturers of dietary supplements varies
widely in quality, species of origin, and country of origin. Only
through reliable testing by a qualified analytical laboratory can
the manufacturer of chondroitin-containing products be certain of
its potency. Unfortunately, this expensive ingredient is widely
available in adulterated form, and may vary in potency more than
perhaps any other ingredient in the largely unregulated health food
industry, ranging from less than 10% to greater than 90% purity.
Below we discuss this problem and what is being done to correct it.
Origin
Chondroitin sulfate is typically produced from cartilage obtained
from cattle, pig, or poultry slaughterhouses, or as a byproduct of
the shark fisheries industry. Shark cartilage is most widely used in
Europe and Japan, while bovine chondroitin is more popular in the
US. The worldwide availability of shark cartilage is rather limited,
and much of the presumable marketed shark chondroitin on the market
today is actually derived from bovine trachea. This true origin of
the chondroitin can only be detected by rather expensive and
detailed chemical analyses. Bovine trachea are widely available, and
produce chondroitin molecules of smaller size, which can be more
easily absorbed by the body. A disadvantage of bovine chondroitin is
the common misconception that BSE, the source of mad cow disease,
may be present. However, this fear is unfounded, as BSE prions can
only occur in proteins, which are absent in good quality chondroitin,
due to the complicated extraction processes.
Certificates do not tell the
whole story...
Chondroitin sulfate varies widely in price. Cheap chondroitin,
commonly referred to as “food grade” chondroitin, is commonly
used for animal supplements, as well as in some low cost human
supplements. This chondroitin consists mostly of molecules which
have similar chemical characteristics to true chondroitin molecules,
but are in fact different and not biologically active. While a
product may claim, for example, to contain 200 mg per tablet, the
actual beneficial chondroitin content could be less than 20 mg.
Pharmaceutical grade chondroitin is typically five to ten times as
expensive as food grade chondroitin, but should contain 90 - 98%
pure chondroitin. Manufacturers of dietary supplements often rely on
the “certificate of analysis” (CofA) of specific lots, and it is
uncommon to verify the CofA concentration by an independent
analytical laboratory. Even when manufacturers do have the source
material tested, there may be no guarantee that the results will be
accurate. The problem is that the analytical method currently
accepted by the US Pharmacopeia (USP), the CPC titration method, is
faulty, and can be easily tricked to give false positive results.
This procedure treats molecules which are chemically related to
chondroitin but are quite useless as true chondroitin. While a more
reliable method is already available, it is optional, as it is not
yet the “official” USP method.
It is quite possible
that a CofA which states that the chondroitin concentration is
>90% by USP, may in fact contain little, if any, real chondroitin.
According to Dr. Ron Peterson of Shuster Laboratories, a renowned US
testing lab, dozens of samples are received every week, mostly of
Chinese origin. Shuster Labs is usually requested to analyze these
samples for chondroitin following USP, i.e. the faulty CPC titration
method. It is quite easy to understand how many producers can still
legally claim high quality, i.e. >90% chondroitin, on their
certificates for food-grade material.
Enzymatic HPLC
To put a halt to this practice, Dr. David Ji of Analytical
Laboratory in Anaheim developed a new analytical method which takes
advantage of the ability of certain specialized enzymes to break
down chondroitin molecules into smaller parts, which can then be
analyzed. Since only true chondroitin molecules are attacked by
these enzymes, it is highly specific, and unaffected by the presence
of chondroitin-like molecules. An added advantage is that the
concentrations of the breakdown products, so-called Type A and Type
C molecules, can yield an indication of the origin of the
chondroitin, land-based or marine, and based on the known A/C ratio.
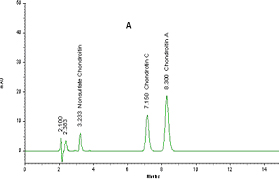 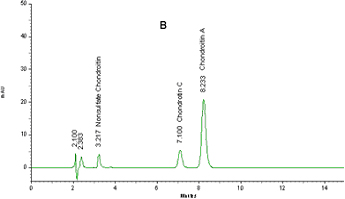
Figure 1: Typical chromatograph of chondroitin from bovine trachea
(A) and from shark (B)
In response to a
growing concern about the quality of chondroitin in today’s
supplement products, the enzymatic HPLC method has been proposed to
replace the CPC titration method as the official USP method of
testing. The method is currently being tested by multiple analytical
laboratories, and following the expected positive outcome of this
study, it is expected to become the new method of choice in circa
2008. Until that time the manufacturer of chondroitin containing
supplements has no choice but to submit samples of the raw material
to a qualified laboratory for analysis by enzymatic HPLC. To avoid
being misled, consumers should put their trust in brand names and
purchase chondroitin-containing human or pet health supplements only
from reputable, high quality manufacturers.
return to previous page |